By Helen Yarimet Lorenzo-Anota and Omar Lozano
Obesity is a complex, multifactorial global health challenge that is difficult to treat. To address this, the scientific community is exploring new alternatives: fields such as biotechnology and nanotechnology are testing treatments based on drugs already approved for other uses.
One such case is Disulfiram, a clinically approved medication for treating alcoholism with anti-inflammatory properties that, in preclinical studies, has been shown to induce weight loss in rats fed a high-fat diet.
Additionally, Disulfiram prevented fat accumulation in the liver and pancreatic hyperplasia, while normalizing insulin response. However, its high instability in the bloodstream limits its clinical application.
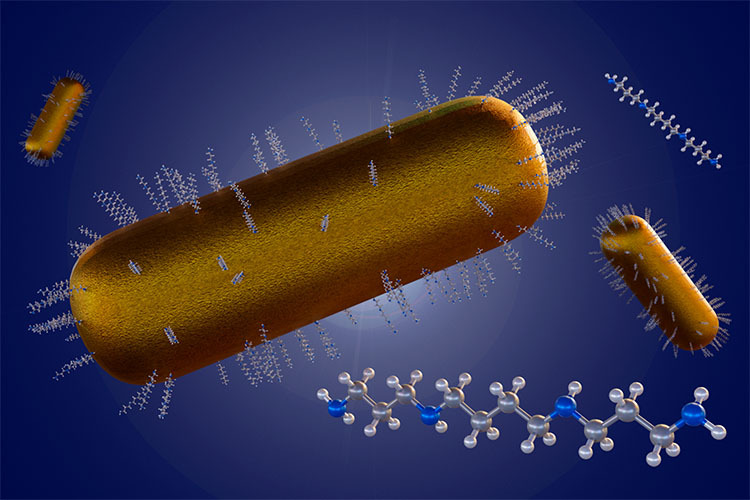
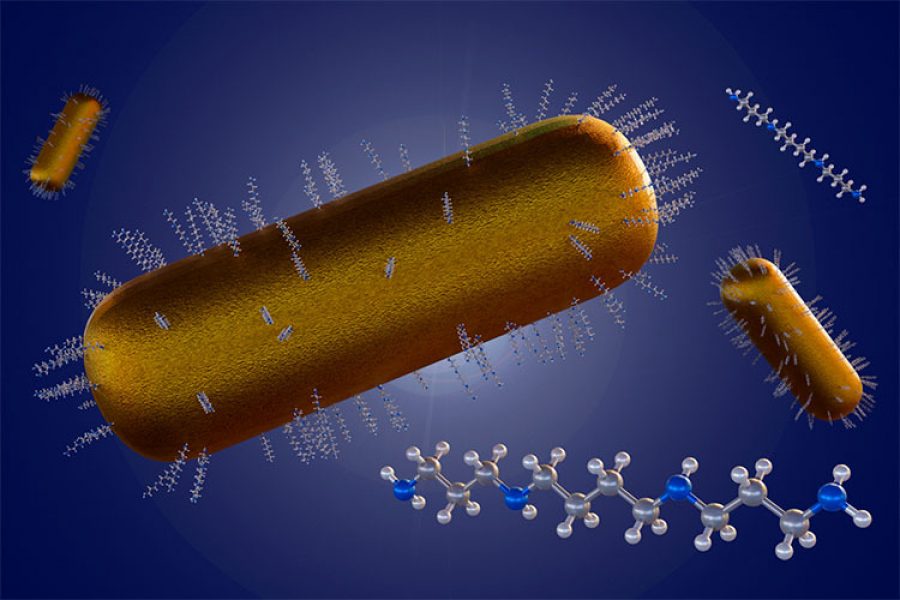
Currently, nanoparticles are being investigated as carriers to transport Disulfiram effectively. Among these, poly-ε-caprolactone (PCL), an FDA-approved biopolymer widely used in biomedical applications due to its high biocompatibility, shows promise.
In other words, PCL encapsulates drugs and transports them within the body. In the study Disulfiram-Loaded Nanoparticles Inhibit Long-Term Proliferation on Preadipocytes, this biopolymer was used to form nanoparticles containing Disulfiram, improving its stability and efficacy.
Biopolymers as Drug Carriers
Our research aimed to optimize particle synthesis by focusing on key properties such as size, homogeneity, and encapsulation efficiency. We found the most promising particles were the smallest: spheroidal in shape and approximately 100 nanometers in size.
Chemical and structural analyses of the nanoformulation showed no alterations compared to the free drug or the nanoparticle alone, suggesting that Disulfiram is physically encapsulated within the polymer matrix.
When evaluating its release profile under simulated conditions (pH 7.4, 37 °C), a controlled release was observed starting within the first 30 minutes, reaching 100% release after four days.
We also analyzed how the nanoparticles interacted with key cells in adipose tissue—an active metabolic organ that not only stores fat but also functions as an endocrine organ by producing hormones and molecules that regulate appetite, metabolism, and the immune system. Dysfunction of this tissue—as occurs in obesity—can lead to metabolic diseases such as type 2 diabetes, insulin resistance, and cardiovascular problems.
In this context, delivering the drug via nanoparticles enabled successful internalization into white adipocytes (fat-storing cells), macrophages (involved in inflammatory responses), and preadipocytes (precursors to new adipocytes)—all cells involved in adipose tissue dysfunction during obesity.
Nanoencapsulation and Biosafety
To understand the cytotoxicity mechanism—meaning the cell damage or death caused by a compound—we evaluated the effect of the encapsulated drug on the mitochondria of preadipocytes. An increase in reactive oxygen species (ROS)—unstable molecules that damage cells and cause oxidative stress—was observed.
This stress activated caspases, enzymes crucial in apoptosis, or programmed cell death. Interestingly, free Disulfiram exhibited higher cytotoxicity than the encapsulated form over the same period, suggesting that encapsulation modulates and potentially reduces the drug’s toxicity.
We compared the efficacy of both drug forms by measuring the proliferation of preadipocytes—cells that can become adipocytes and contribute to obesity. After 24 hours of treatment, cells were allowed to grow for 10 days to observe colony formation. Both treatments similarly reduced cell proliferation, demonstrating comparable efficacy.
These findings suggest that nanoencapsulation of Disulfiram could improve its bioavailability and safety profile, while demonstrating potential anti-obesogenic effects. Beyond confirming its cytotoxicity in preadipocytes, this study opens the door to developing therapeutic strategies targeting adipose tissue in obesity.
Next steps include progressing to more complex models such as three-dimensional cultures and animal studies. If these results are confirmed, they would establish a strong proof of concept to move forward toward clinical trials in humans.
References
- Lorenzo-Anota HY, Gómez-Cantú JM, Vázquez-Garza E, Bernal-Ramirez J, Chapoy-Villanueva H, Mayolo-Deloisa K, Benavides J, Rito-Palomares M, Lozano O. Disulfiram-Loaded Nanoparticles Inhibit Long-Term Proliferation on Preadipocytes. Int J Nanomedicine. 2024 Dec 10;19:13301-13318. doi: 10.2147/IJN.S467909. PMID: 39679252; PMCID: PMC11645963.
- Kaluzynski K, Pretula J, Lewinski P, Kaźmierski S, Penczek S. Synthesis and properties of functionalized poly(ϵ-caprolactone); chain polymerization followed by polycondensation in one pot with initiator and catalyst in one molecule synthesis and molecular structures. Macromolecules. 2022;55(6):2210–2221.
- El Yousfi R, Brahmi M, Dalli M, et al. Recent advances in nanoparticle development for drug delivery: a comprehensive review of polycaprolactone-based multi-arm architectures. Polymers. 2023;15(8):1835.
- Johansson B. A review of the pharmacokinetics and pharmacodynamics of disulfiram and its metabolites. Acta Psychiatr Scand. 1992;86 (S369):15–26.
- Cvek B. The promiscuity of disulfiram in medicinal research. ACS Med Chem Lett. 2023;14:1610–1614.
- Choe SS, Huh JY, Hwang IJ, Kim JI, Kim JB. Adipose tissue remodeling: its role in energy metabolism and metabolic disorders. Front Endocrinol. 2016;7(APR).
- Sakers A, De Siqueira MK, Seale P, Villanueva CJ. Adipose-tissue plasticity in health and disease. Cell. 2022;185(3):419–446.
- Zatterale F, Longo M, Naderi J, et al. Chronic adipose tissue inflammation linking obesity to insulin resistance and type 2 diabetes. Front Physiol. 2020;10.
- Rohm TV, Meier DT, Olefsky JM, Donath MY. Inflammation in obesity, diabetes, and related disorders. Immunity. 2022;55(1):31–55.
- Labet M, Thielemans W. Synthesis of polycaprolactone: a review. Chem Soc Rev. 2009;38(12):3484–3504.
- Homaeigohar S, Boccaccini AR. Nature-derived and synthetic additives to poly(ɛ-Caprolactone) nanofibrous systems for biomedicine; an updated overview. Front Chem. 2022;9.
.
Author
Helen Yarimet Lorenzo Anota. Ph.D. in Immunobiology, UANL. Currently, a postdoctoral researcher at the Institute for Obesity Research, Bioengineering and Medical Devices Unit, at Tecnológico de Monterrey. Her research focuses on developing novel systems for the efficient delivery of anti-obesogenic molecules in in-vitro models, particularly on cells derived from adipose tissue, including preadipocytes, adipocytes, and immune cells. Member of the National System of Researchers, Level I, since 2023.
Omar Lozano. Professor-Researcher at the Institute for Obesity Research and the School of Medicine and Health Sciences at Tecnológico de Monterrey. His research focuses on developing advanced, efficient, and safe materials for delivering therapeutic and diagnostic compounds for cardiometabolic diseases. He has written 46 scientific and outreach articles (h-index: 20), 4 patents, and 2 book chapters. He is a member of Mexico’s National System of Researchers, Level 2, and a Chargé de Recherche in Belgium.