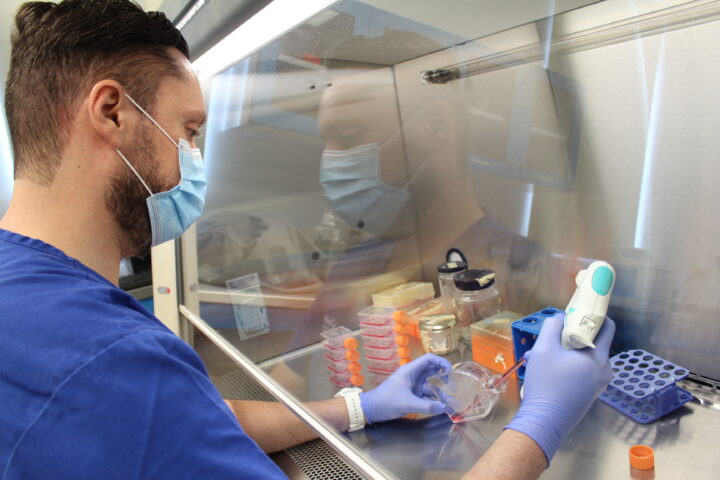
The country is generating applied science and clinical success stories. But without early-stage funding and stronger coordination across sectors, those advances may never translate into real social impact.
Medical innovation in Mexico is at a turning point, according to experts in industry, academia, and regulatory bodies. However, strategic changes are still needed to accelerate innovation, retain Mexican talent, and increase coordination among the key players in the scientific ecosystem.
At the TecScience Summit 2026, panelists examined the structural challenges facing the country’s health innovation ecosystem. Mexico has the talent and the scientific infrastructure, but critical obstacles remain before the country can become a global reference in medical innovation.
Mexico’s Medical Innovation Ecosystem
For Sonia Mayra Pérez Tapia, the current situation is clear: “There is no innovation in Mexico.” The founder and executive director of the Biotherapeutic Development and Research Unit at the National Polytechnic Institute issued a warning: “Companies come and conduct clinical trials in Mexico, but in terms of us generating disruptive innovations, we are not doing that.”
She was also the first to soften her stance: there is incremental innovation, talent, and ideas in the laboratory. The obstacle, she explained, arises in the transition from lab to market, a process that demands regulatory expertise and access to financing. That gap is something that current education systems have not addressed.
Clinical Trials and Regulatory Development: From the Lab to the Market
Gerardo García Rivas, director of Translational Research at TecSalud, offered a counterargument from his own experience. His team has spent eight years developing a cannabidiol-based formulation for the treatment of myocarditis, with trials involving 109 patients in Mexico, the United States, and Brazil.
“We were able to get from a molecule in a culture dish in Mexico to the first human test subjects,” he said. For García, the Mexican ecosystem is beginning to show results, though he acknowledged there are still plenty of opportunities to grow.
José Guillermo Sander Padilla, senior manager of Clinical Innovation at Silanes Laboratory, presented the most concrete case of industrial success: the first innovative Mexican medication approved by the Food and Drug Administration (FDA) for commercialization in the United States. He highlighted that the key to reaching this milestone was early clinical development planning, which has allowed his company to achieve a success rate of 64.6 percent in new regulatory entities, compared to the 2 percent reported in global data.
Abraham Israel Villarreal Castellanos, coordinator of clinical trials at COFEPRIS, did not deny that innovation exists in Mexico. From his perspective, the obstacles go beyond innovation itself. “What we’re missing is for it to translate into social well-being. A lot of times it stays in academia, in a development plan, and never reaches where it needs to go,” said Villarreal.

(Photos: Everth Bañuelos)
The Problem Isn’t Talent — It’s the System
On the question of scientific expertise, there was consensus. Mexico produces quality researchers, has access to centers of excellence, and benefits from a privileged geographic position next to the world’s largest pharmaceutical market. The problem, the panelists agreed, is systemic: what’s missing is for academia, industry, and regulatory bodies to work in a coordinated way rather than in parallel, as they do now.
COFEPRIS is redesigning its processes to simplify regulations and align more closely with agencies such as the FDA, the Mexican Accreditation Entity, and Health Canada. They are also looking to reduce authorization times without compromising the safety of patients participating in clinical trials.
On the academic side, García explained how attitudes toward industry are changing. Fifteen or twenty years ago, he said, it was unthinkable for a cardiovascular physiology researcher to work alongside a company. Today, that line has blurred, and he sees an opportunity to share academia’s scientific knowledge. “It’s exactly what industry and regulatory agencies need in order to know what is possible,” he said.
This shift in perspective also applies to researchers, who often focus on scientific questions and lose sight of the social impact that the pharmaceutical industry is looking for.
How to collaborate to generate real impact
When asked how they can better collaborate to generate real impact, Pérez proposed three concrete actions from academia: teach regulatory norms in the early stages, give students a clear picture of the costs involved in developing health products, and design research programs with the country’s specific needs in mind. “We can do this from academia in a very targeted way,” said Pérez.
From the industry side, Sander explained that all three actors need to change their perspective. Academia cannot simply focus on filing patents without developing products. Industry cannot rely on money and wait for innovation to come to them. And regulatory bodies must find ways to integrate those actors into the process from the start.
Regulatory agencies must also learn to trust the research ecosystem. Rather than repeating trials or research processes when they reach COFEPRIS, the ideal would be to build on the work already done.
“In the end, everything that comes out of academia and industry needs to be regulated, one way or another,” said Villarreal. That is why it is important to work together with those actors from the beginning.
Retaining Scientific Talent: Mexico’s Structural Challenge
Despite the obstacles, the panelists agreed that Mexico has competitive advantages that other countries with similar economies do not. The country’s geographic position, bordering the world’s largest pharmaceutical market, represents an opportunity that has not yet been fully seized.
This, combined with Mexico’s unique biodiversity, strong scientific foundation, and a national pharmaceutical industry that—as in the case of Silanes Laboratory—is already capable of competing at international standards, makes for a compelling base to build on.
The challenge is creating the conditions to retain the talent coming out of Mexican universities: more early-stage financing, stronger connections between academia and industry, and more spaces where scientists can build their careers without having to leave the country.
The panel’s closing message was that Mexico is not at the starting point, but it has not yet reached a finish line either. It is at an inflection point, and the decisions that academia, industry, and regulators make will determine whether the country becomes a global reference in medical innovation or continues exporting talent instead of products.
Did you find this story interesting? Would you like to publish it? Contact our content editor for more information: marianaleonm@tec.mx